How can equitable treatment of myeloma be secured in the future?
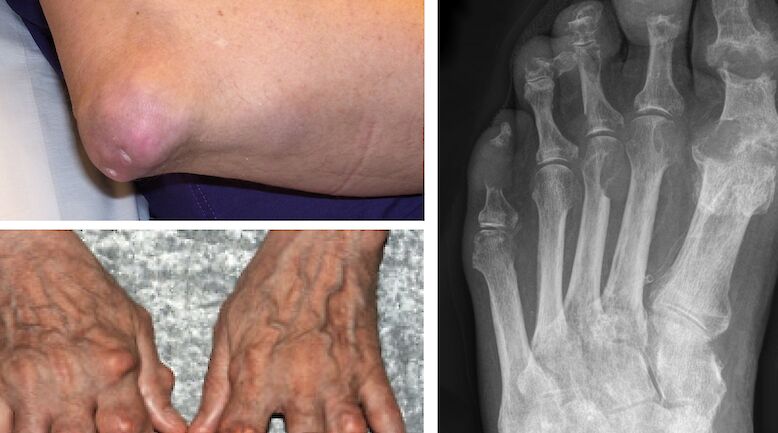
Multiple myeloma is a neoplastic disorder characterized by the proliferation of a single clone of plasma cells derived from B-cells. This clone grows in the bone marrow leading to skeletal destruction. Myeloma constitutes approximately 1% of all cancers and 10% of haematological malignancies. Patients are on average 65–70 years old at diagnosis.
The annual incidence of myeloma remains stable, but the number of patients living with myeloma is increasing due to the development of novel therapies. Despite improved treatment options, myeloma remains practically incurable with current therapy, and is characterized by multiple relapses. It typically recurs with a more aggressive disease course after each remission. Thus, treatment is always planned individually for each patient considering the advantages and disadvantages of the previous treatment.
The growing prevalence of myeloma is a positive challenge for the healthcare system, as it indicates that the treatment of myeloma has advanced and patients are surviving longer than before. To secure the necessary resources in the future, issues related to the management of myeloma must be identified, and solutions actively sought. One of the concerns is a significant shortage of clinical haematology specialists, which may compromise equitable treatment. In addition, the cost of myeloma treatment is increasing, mainly due to the higher cost of novel therapies. Various risk-sharing agreements strive to manage these costs for both in-hospital and outpatient usage. Even then, hospital budget considerations can limit the use of in-hospital drugs. Importantly, clinical trials provide valuable early-stage experience with investigational new drugs and concrete savings for hospital drug budgets. As a result, their implementation in parallel with routine treatment should be enabled. In addition to clinical trial participation, it is important to develop tools to monitor treatment efficacy and safety also in a real-world setting.
Raija Silvennoinen, Pekka Anttila, Juha Lievonen, Marja Sankelo, Marjaana Säily, Anu Partanen, Mervi Putkonen
Raija Silvennoinen
M.D., Ph.D., Researcher
Department of Hematology, Comprehensive Cancer Center, University of Helsinki, and HUS Helsinki University Hospital
raija.silvennoinen@helsinki.fi